The morning after pill can be abortifacient and women need to know the truth. I read the article by Isabel Stabile, published on TMIS of Sunday, 6 December, and as a researcher and gynecologist in the field, I must answer in the interest of correct consumer information to the Maltese citizens.
I base my reply quoting the Official Assessment Reports on EllaOne by the European Medicines Agency (EMA) itself. It is pertinent to note that we are speaking of two very different drugs mentioned as Morning after pill, each having a completely different mode of action. These are Levonogestrel (morning after pill) and Ella One (five days after pill).
From the studies presented, it is clear that Levonogestrel (LNG) is unable to prevent or delay ovulation; however, it leads to a shortened or inadequate corpus luteum that does not allow the endometrium to prepare for implantation. The diagram below, from a study by Brache et al, confirms that compared to the placebo, Levonogestrel does not prevent ovulation. The data is presented and shared by EMA itself in its EPAR (European Public Assessment Report) on EllaOne page 9 and updated yearly; last update being on 20 May.
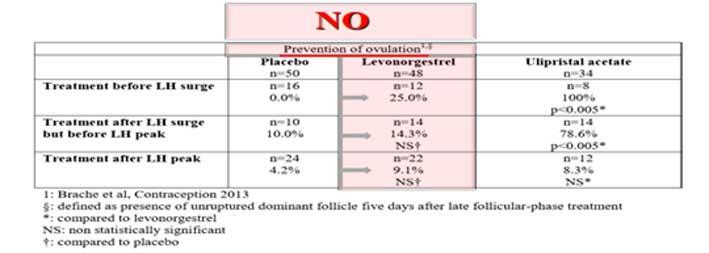
The EMA shows officially that LNG does not work by affecting ovulation and is placebo-like in the fertile days, when fertilization is possible. In spite of this, the EMA presents LNG as an ovulation-inhibitor. Why?
EllaOne (ulipristal acetate, UPA) can prevent implantation and also terminate ongoing pregnancies. “Ulipristal acetate prevents progesterone from occupying its receptor, thus the gene transcription normally turned on by progesterone is blocked, and the proteins necessary to begin and maintain pregnancy are not synthesized.” (EMEA 2617/87/2009, Page 8: 2.3). On page 10 of the same document, it goes on to say that: “The ability of Ulipristal Acetate (UPA) to terminate pregnancy was investigated. Ulipristal, mifepristone and lilopristone were approximately equipotent” further confirming that it can terminate an ongoing pregnancy.
The science and the marketing of the morning after pill (MAPs) are diametrically opposed. The consumer can easily be misled into thinking that the MAPs are “normal” contraceptives when in fact they can be and are anti-implantation drugs and EllaOne can likely interrupt ongoing pregnancies. Women who would be reluctant to take any medicinal that could endanger their embryo’s life should have access to the correct information and biological processes taking place in their body. The law in Malta protects the embryo from conception. Professionals can also have a conscientious objection to referral and/or supply of such medicinals.
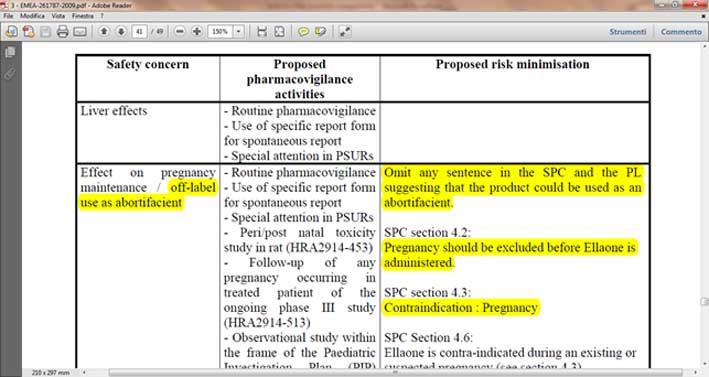
In her article, Stabile also emphasised the relevance of the SPC. Have a look at the image below taken from page 41 of the same 2009 EMA Assessment Report: the highlighted sentences: “Omit any sentence in the SPC and the PL suggesting that the product could be used as an abortifacient”, “Pregnancy should be excluded before EllaOne is administered” and “Contraindication: Pregnancy”, speak volumes.
Furthermore, the European Medicines Authority in the Assessment Report EMA/73099/2015 on page 35, reports that during the evaluation process of the ellaOne registration dossier, the company, HRA-Pharma, was “requested to study any potential off-label use of ellaOne, in particular during pregnancy, possibly as an abortifacient. No such clinical studies were performed with Ulipristal-Acetate and it is therefore left as an unknown, whether it is possible to use it for abortion”. This notwithstanding, at the end of the 2015 AR, EMA presented ellaOne once again as an anti-ovulatory drug and made it “not subject to medical prescription”.
EllaOne does not prevent ovulation in the most fertile days and yet over 80% of expected pregnancies do not appear clinically. The drug does not allow endometrial maturation, so necessary for the embryo to implant. This leads to the embryo losing his or her life.
EllaOne and Levonogestrel are being sold over the counter and without prescription to young adults and women who think that they are using contraceptives that are safe. No advice is obligatory as to the side effects that these medicinals can have on their health, especially with recurrent use of EllaOne. In fact, Esmya, a medicine containing the same active ingredient, ulipristal acetate, has been withdrawn from clinical use due to a direct relationship between UPA and severe liver injuries, including liver transplantation. A case of severe DILI (Drug Induced Liver Injury) occurred after an UPA consumption, not greater than the dosage in one-two tablets of EllaOne, and EllaOne, unlike Esmya, can be self-administered repeatedly, without any medical surveillance.
The scientifically correct information regarding the mechanism of action of these drugs must be available to our politicians, decision-makers, doctors, chemists and other professionals and last but not least to the women consuming these pills. Their rights and freedom of informed choice are being deliberately thwarted. Women are being deceived. They are being informed that ovulation will be prevented after taking Emergency Contraceptives, but, on the contrary, they can ovulate and can conceive, but their offspring dies as these drugs inhibit implantation. Healthwise they are completely unaware of the severe drug induced liver injury (DILI) potentially due to Ulipristal acetate.
For further information on toxicity and on the mechanism of action, please see, respectively: Ulipristal Acetate and liver-injuries: while Esmya is revoked, EllaOne is allowed in repeated self-administrations possibly exceeding UPA toxic-dosing with Esmya. Mozzanega B. J Hepatol 2020 Nov. 30; S0168-8278(20)33828-9. doi: 10.1016/j.jhep.2020.11.041.
UPA and LNG in emergency contraception: the information by EMA and the scientific evidences indicate a prevalent anti-implantation effect. Mozzanega B, Nardelli GB. Eur J Contracept Reprod Health Care 2019; 24(1):4-10. doi: 10.1080/13625187.2018.1555662. Epub 2019 Jan. 18
Further scientific papers are available on http://www.sipre.eu in Italian or English
Bruno Mozzanega, Dept SDB (Woman’s and Child’s Health), University of Padua (I)